III Isotopes
Determined by the number of neutrons.Ex. C12 & C14*C14 is used in carbon dating its half life is 5,730yrs.*Half Lives
IV Ions-Electrically charge atom or molecule.
-This happens when Protons do not equal Electrons.-Important ones: H+, OH-, K+, Cl-, Na+,Ca2+ .
-Some cells may not let any ions pass through
Ex. Cystic Fibrosis, which creates a lack in sodium and chlorine.
V Bonding
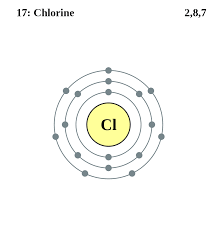
A. Ionic -these bonds form between oppositely charged ions.Ex. Na+ (1 valence e-) and Cl- (7 valence e-)
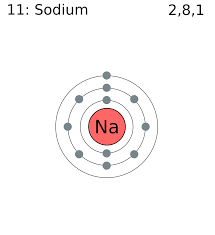 .
.
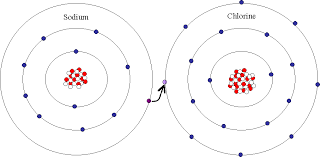 -Bonding takes place and chlorine takes an electron from sodium completing its outer shell and completing sodium's shell. Both receive an octet and together they make NaCl.
-Bonding takes place and chlorine takes an electron from sodium completing its outer shell and completing sodium's shell. Both receive an octet and together they make NaCl.
2types: 1.Non-polar- when electrons are shared equally.
Ex. H2 H-H Single Bond. O2 O=O Double Bond. N2 Triple bonded!
No comments:
Post a Comment